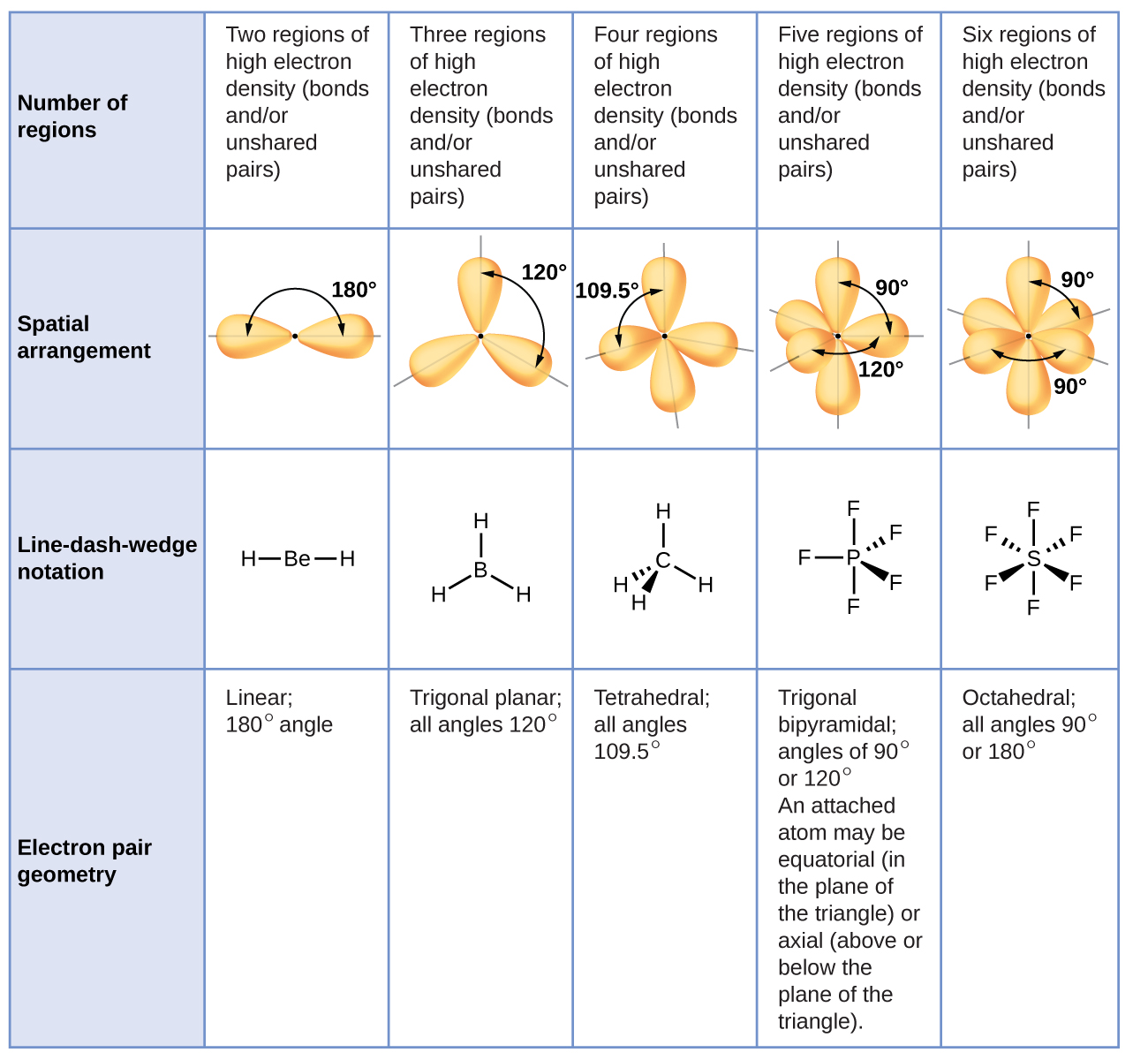
Thus the chart clearly shows that the molecular geometry of PCl3 is a trigonal pyramid.Īlso, the electron geometry of PCl3 is tetrahedral. 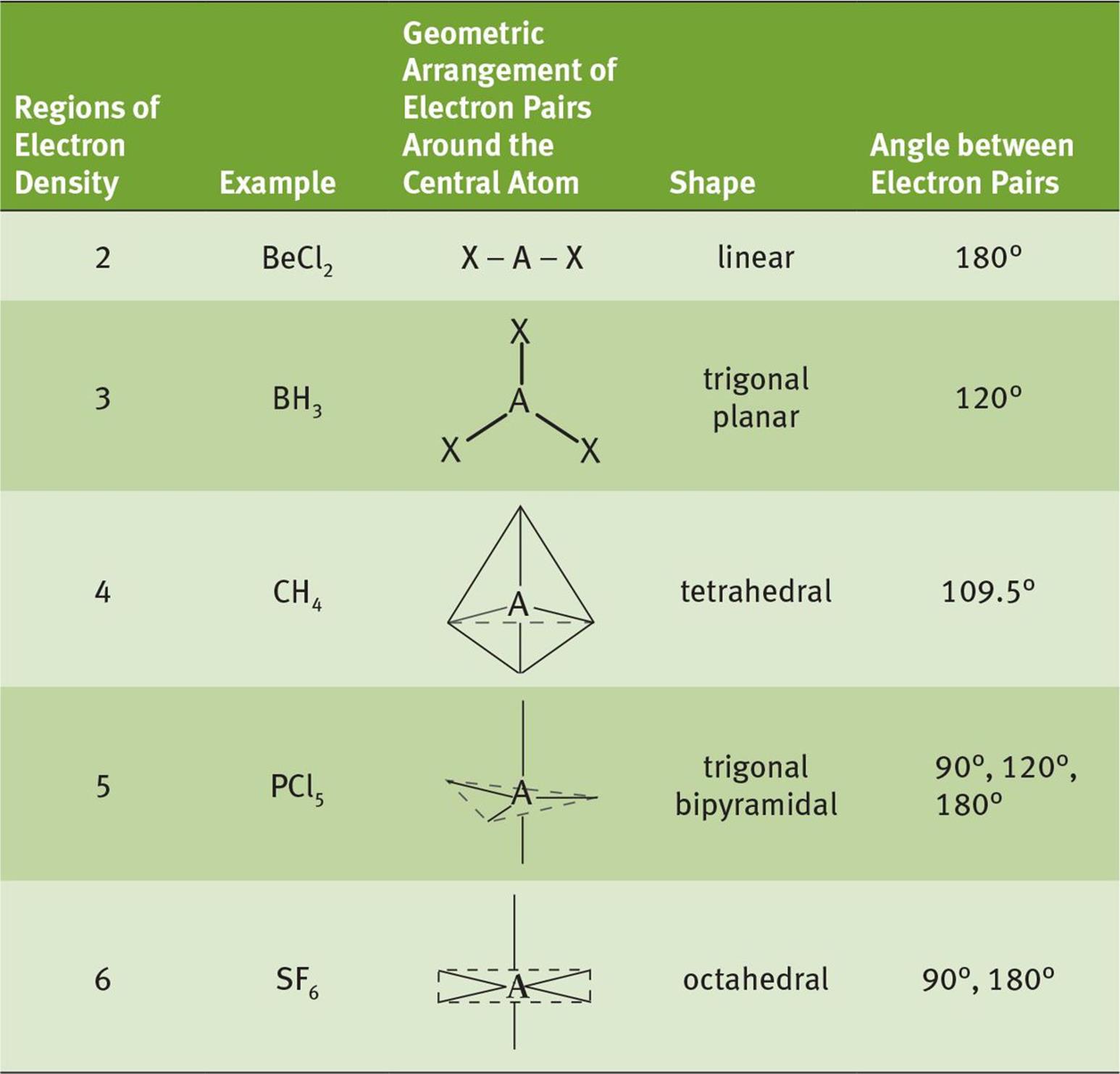 
PCl3 has 1 lone pair and 3 surrounding atoms. Now PCl3 is a AX3E type molecule where, A = central atom, X = surrounding atom, E = lone pair. The below-attached VSEPR chart gives us an idea about that The molecular geometry of any compound can be determined easily from the VSEPR theory. The molecular geometry of PCl3 is a trigonal pyramid. These two concepts clearly explain the Sp3 hybridization of PCl3. Here, V (valence electron of central atom) = 5Īs it is a neutral compound thus C and A will be 0 Let’s apply this formula to find the hybridization of PCl3 Not only PCl3, but this formula can also be used to find the hybridization of any compound. This concept very well explains the hybridization of PCl3 which is sp3.Īnother simple formula can also give us the hybridization of PCl3. With that 2 lone pairs are present on the phosphorus atom. We can clearly see from the lewis diagram that in PCl3, phosphorus is forming three sigma bonds with 3 chlorine atoms. We can understand this concept with the help of either the concept of bonding or by using a simple formula.įirst, let’s understand the bonding part. The hybridization of a molecule generally gives us ideas about the mixing of orbitals in a compound. Let’s move to the hybridization of phosphorus trichloride. Every atom should be in its least possible formal charge,Īlso, every atom in the lewis structure of PCl3 is fulling octet, as they are having 8 electrons each after sharing. Lastly, to ensure the lewis structure is completely correct, we need to check the octet of every atom and also their formal charge. So, 20 electrons are remaining which needs to be given around the atoms.Īfter fulfilling the electrons we can see the final lewis structure of PCl3. The next job is to fulfill the octet of the atoms with the remaining electrons.įor the single bonds in the skeletal structure, 6 electrons are used. The skeletal structure is drawn with single bonds only. Now to draw the final lewis structure of PCl3, we need to start from the skeletal structure. The central atom is basically the atom with the highest number of bonding sites. To draw the lewis structure, first of all, we need to sum up the valence electrons of all the atoms. The lewis structure of PCl3 can be explained as follows : Now let’s move on to the lewis structure of PCl3. The calculation for formal charge can be done using the formula given below:. At the end check that all the atoms are in their lowest possible formal charge.Check if there is a requirement of multiple bonds for fulfilling the octet rule of all atoms.Always begin with the electronegative atoms and then the electropositive ones. Complete the octet of the atoms with the electrons left.Create a skeleton containing only single bonds.Identify the central atom the atom with the highest bonding sites.A ‘+’ sign means losing electrons and ‘-‘ means gaining. Sum up the total number of valence electrons in the molecule.Now to simplify the process of drawing a lewis structure I have jotted down the steps in bullets:. The lewis structure of any compound helps us to find out the number of bonds, types of bonds, and the interaction between the atoms in a compound. So without any further ado let’s jump into these topics in detail!Ī lewis structure mainly focuses on fulfilling the octet of the atoms in a compound. From being used in reactions to the production of something PCl3 can be used for different needs.īut before going into reactions involving PCl3, it is very necessary to know about the structure, hybridization, and bonding of this liquid. Like any other compound, this also has immense use in different fields. So to avoid that continuous removal of phosphorus trichloride is mandatory. Phosphorus trichloride can be produced by the treatment of chlorine with a refluxing solution of white phosphorus in PCl3.Īlso, there is a high chance of PCl5 formation in this process. 
So, PCl3 is a covalent molecule because here equal sharing of electrons forms the bond between phosphorus and chlorine. Is PCl3 polar or nonpolar? Don’t worry, the answer is simple! PCl3 is a polar molecule because of its geometry and difference in electronegativity between the 2 atoms.Īgain another question like, whether PCl3 is ionic or covalent, can pop up in your mind. Now there can be questions about the polarity of this compound. The melting point and boiling point of this compound are -93.6℃ and 76.1℃ respectively. The molar mass of this compound is 137.33 g/mol. PCl3 is a toxic liquid with an unpleasant smell. Phosphorus trichloride with a chemical formula PCl3 is a yellow fuming liquid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
